Amblyopia Treatment Using a Portable Display Device

Publications
- The iPod binocular home-based treatment for amblyopia in adults: efficacy and compliance. Hess RF, Babu RJ, Clavagnier S, Black J, Bobier W, and Thompson B. Clin Exp Optom. 2014 Sep;97(5):389-98.
- The measurement and treatment of suppression in amblyopia. Black, J. M.; Hess, R. F.; Cooperstock, J. R.; To, L.; and Thompson, B. Visualized Experiments, 14(70). 2012.
- An iPod treatment for amblyopia: An updated binocular approach. Hess, R. F.; Thompson, B.; Black, J. M.; Maehara, G.; Zhang, P.; Bobier, W. R.; To, L.; and Cooperstock, J. R. Optometry, 83(2):87-94. February 2012.
- A game platform for treatment of amblyopia. To, L.; Thompson, B.; Blum, J.; Maehara, G.; Hess, R.; and Cooperstock, J. R. IEEE Transactions on Neural Systems and Rehabilitation Engineering, 19(3):280-289. June 2011.
In addition, our prototype based on a head-mounted display (HMD) is being used by other research groups, including Glasgow Caledonian University, to explore this treatment-via-gaming approach. Further details are available from the Investigative Ophthalmology & Visual Science publication, “An Exploratory Study: Prolonged Periods of Binocular Stimulation Can Provide an Effective Treatment for Childhood Amblyopia”.
Background
- amblyopia (or “Lazy Eye” syndrome) affects ~2% of population
- generally diagnosed in childhood and treated by patching the “good eye”
- lifetime risk of loss of vision if untreated >1.2%
- patching carries social stigma, resistance to use
A New Approach
- modified Tetris game implemented on a portable gaming device
- based on patented McGill treatment method
- takes advantage of 3D display technology to provide differential contrast to the two eyes
- currently integrating a head-tracking feature using front-facing camera to prevent the patient from “cheating”
Our Treatment Approach
- present different stimulus to the two eyes at different contrast ratios:
- weak eye receives stronger signal and thus engages in visual process
- over time, contrast ratio between the eyes is reduced
- both eyes must see the content to play the game effectively
- therapeutic benefit from gaming
- children and adults actually encouraged to play video game to improve their vision
More Details
Amblyopia is a visual disorder in which information from the good (fellow) eye dominate and suppress the information from the amblyopic eye. An amblyopic person only sees a monocular view of the environment, often without the depth information, which can lead to perceptual problems. Amblyopia is caused by abnormal development in the visual cortex, and may be present even when there is no physical defect at the amblyopic eye. Unlike myopia, it cannot be corrected by an optical lens.
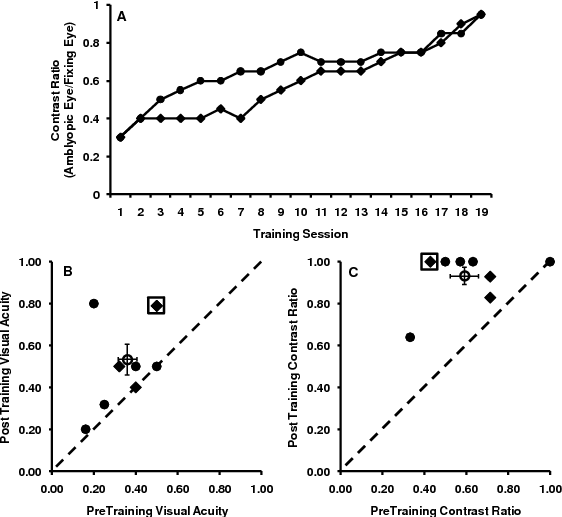
We have developed a prototype device for take-home use that can be used in the treatment of amblyopia. The therapeutic scenario we envision involves patients first visiting a clinic, where their vision parameters are assessed and suitable parameters are determined for therapy. Patients then proceed with the actual therapeutic treatment on their own, using our device, which consists of an Apple iPod Touch running a specially modified game application. Our rationale for choosing to develop the prototype around a game stems from multiple requirements that such an application satisfies. First, system operation must be sufficiently straightforward that ease-of-use is not an obstacle. Second, the application itself should be compelling and motivate use more so than a traditional therapeutic task if it is to be used regularly outside of the clinic. This is particularly relevant for children, as compliance is a major issue for current treatments of childhood amblyopia. However, despite the traditional opinion that treatment of amblyopia is only effective in children, our initial results add to the growing body of evidence that improvements in visual function can be achieved in adults with amblyopia.
Prototype Device



Results